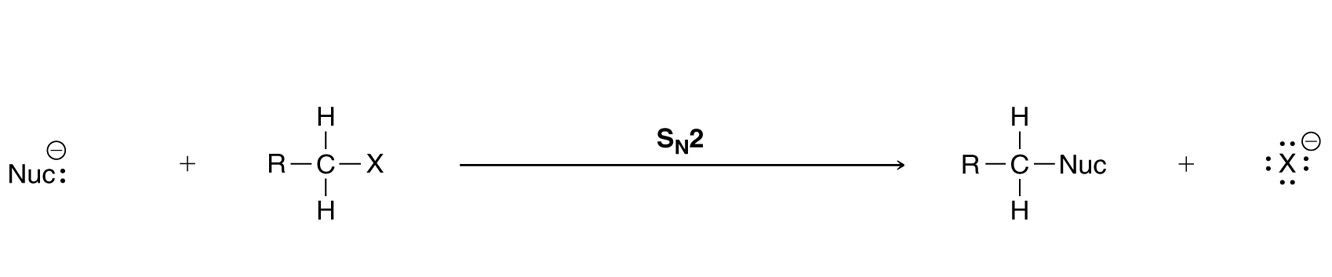
Directions:
Click the "Begin" button below to access an interactive drag and drop version of this graphics, which will help reinforce the concepts you have been studying.
Directions:
Click the "Next" button below to access an interactive drag and drop version of this graphic, which will help reinforce the concepts you have been studying.
Hide Graphic
Reset
View Graphic
Begin